|
5/27/2023 0 Comments Metallic bonds and dispersio force 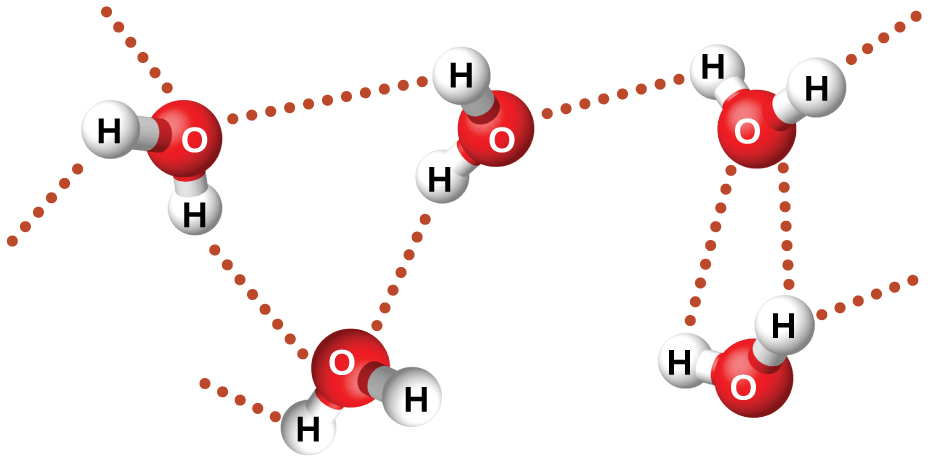
Sodium is a metal therefore, it has a very low electronegativity (0.9) compared to Chlorine (3.0). An atom with high electronegativity can attract electrons from an atom with low electronegativity to form an ionic bond.įor example, sodium chloride has an ionic bond between sodium ion and chloride ion. Hence, electronegativity gives a measurement of the atoms’ affinity for electrons. The electronegativity of the atoms in an ionic bond influences the strength of the electrostatic interactions. Accordingly, ionic bonding is the attraction force between these oppositely charged ions. There are electrostatic interactions between these ions. Side by Side Comparison – Ionic Bonding vs Metallic Bonding in Tabular FormĪtoms can gain or lose electrons and form negative or positive charged particles respectively. This occurs by forming ionic bonds, covalent bonds or metallic bonds. Thus, each atom can achieve a noble gas electronic configuration. These atoms tend to react with each other to become stable. Most of the atoms have less than eight electrons in their valence shells (except the noble gases in group 18 of the periodic table) therefore, they are not stable. The key difference between ionic bonding and metallic bonding is that the ionic bonding takes place between positive and negative ions whereas the metallic bonding takes place between positive ions and electrons.Īs American chemist G.N.Lewis proposed, atoms are stable when they contain eight electrons in their valence shell. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
